How Does Stress Affect Genitourinary System Peer Reviewed
Introduction
Stress is an organism's attempt to adequately respond to either internal or external threats or injuries. It refers to any concrete or psychological challenge that threatens or has the potential of threatening the equilibrium of an organism'south internal milieu (homeostasis) (1–three). Such challenges could be life events, situations, emotive feelings, and interactions that adversely affect the individual'south wellbeing or trigger perceived harmful responses. Psychosocial stress stimuli comprise life experiences that include changes in personal life and relationships, occupation, housing, family unit limerick, and domestic violence necessitating adaptive survival behaviors/responses from the afflicted private (4, 5). Stress stimulates the hypothalamic-pituitary-adrenal (HPA) axis, which eventually leads to elevated levels of cortisol. As a downstream effector of the stress-induced neuroendocrine response, cortisol exerts global effects in the torso to maintain homeostasis and enhance the organism'southward capacity to respond to and grapple with physical and emotional stresses (6). For example, it primes the organism for "fight or flying" past promoting energy metabolism via glycogenolysis, gluconeogenesis, proteolysis and lipolysis, also as regulates several immune and inflammatory responses (7). Furthermore, it increases blood force per unit area, has diverse bone furnishings, elicits both positive and negative effects on cell growth, and facilitates apoptosis in certain cell types, including sure neuronal cells (8). Normal immune function may exist impaired or dysregulated by exposure to chronic stress through the HPA axis and the sympathetic-adrenal-medullary (SAM) axis, resulting in the chronic production of glucocorticoid hormones and catecholamines (9)
Cortisol is a steroid (glucocorticoid) hormone produced by the zona fasciculata of the adrenal cortex within the adrenal gland. It is released by the adrenal glands as office of the fight-or-flight machinery in response to stress or fear, just has been described every bit public health's foremost enemy (10). Its functions include the modulation of increased claret sugar through gluconeogenesis and induction of insulin resistance (xi, 12), metabolism of fatty, poly peptide, and carbohydrates, growth and reproduction, immune suppression (13), sodium-potassium transport (fourteen, 15), cognition and retentivity (13, 16), and the regulation of bone formation (17).
The release of cortisol is regulated by the HPA axis via the secretion of corticotropin-releasing hormone (CRH) by the paraventricular nucleus (PVN) of the hypothalamus, which stimulates corticotrophs in the inductive pituitary to secrete adrenocorticotropic hormone (ACTH, corticotropin), which travels through the bloodstream to the adrenal cortex. ACTH stimulates the synthesis of cortisol and other glucocorticoids. Cortisol ultimately inhibits the HPA neuroendocrine response via a negative feedback mechanism to restore a steady state (13, 18). Cortisol has a circadian rhythm with everyman levels at midnight and superlative levels (~399 nmol/l) in the morning (around 8:30) (xix).
Hitherto, most studies have attributed the pathogenesis of stress-related vaginal dysbiosis solely to impaired immune function and loss of Lactobacillus species dominance (9, 20), while others have only enumerated the use of corticosteroids as a factor associated with Bacterial vaginosis (BV). Nevertheless, the mechanisms by which cortisol, the classical stress hormone, modulates the estrogen-induced degradation and accumulation of glycogen in the vaginal epithelium and the implications for maintenance of vaginal homeostasis has received little attention. Due to the importance of the association betwixt vaginal glycogen, Lactobacillus species authorisation and low pH for the reproductive health of women, i.e., reducing the risk of sexually transmitted infections (STIs), BV, and preterm labor (21), this review examines the vaginal glycogen response induced by estrogen and the potential repressive role of cortisol.
Literature Search
With the use of words and phrases including (but not limited to) "stress and vaginal health," "stress and bacterial vaginosis," "stress and vaginal infection," "stress and immune role," "stress and vaginal Lactobacilli," "stress, infection and preterm birth," "corticosteroids, cortisol and bacterial vaginosis," "cortisol and vaginal infection," "cortisol and vaginal Lactobacilli," "cortisol and vaginal glycogen," "estrogen, vaginal glycogen and Lactobacillus dominance," a comprehensive search was conducted for scientific, peer-reviewed and published original research articles involving both humans and animal models, equally well equally review articles in PubMed/MEDLINE and Web of Science databases betwixt December 2017 and August 2018. In gild to comprehensively review relevant literature relating to psychosocial stress, cortisol levels, and maintenance of vaginal health, publication dates were not restricted. Manufactures not written in English language or including other hormones not straight involved in the HPA or SAM stress axes and vaginal microbial composition (east.thou., insulin), were not included.
Normal Vaginal Microenvironment
The human vagina is not only a passage for sperm, menstruation, and the baby but also a particularly versatile organ with a protective epithelium and a rich diverse microbial landscape (22). The vaginal microbiome comprises of a stratified squamous non-keratinized epithelium overlaid by a mucin-rich mucus layer and provides an attachment surface for the commensal and near dominant lactic acid producing Lactobacillus species. The nigh frequently identified species of Lactobacillus are L. crispatus, L. jensenii, 50. gasseri, and L. iners (23, 24). Other bacteria endogenous to the normal vaginal microenvironment albeit with low virulence capacity include Gardnerella, Prevotella, Fusobacterium, Atopobium, Streptococcus, Staphylococcus, Peptostreptococcus, Porphyromonas etc. These potentially pathogenic bacteria are kept fallow by the acidic milieu (pH 3.5–4.5) created by Lactobacilli amongst other protective mechanisms including production of lactic acid (~110 mM) (25, 26), hydrogen peroxide (H2O2), antimicrobial peptides and by competitive exclusion i.e., physically preventing the attachment of pathogens to vaginal epithelium (22, 27, 28). The commensal and potentially harmful vaginal microorganisms, their genes and products collectively grade the vaginal microbiota that dwell in a regulated mutualistic relationship with the host vaginal epithelium to class the microbiome (29).
The vaginal microbiota in childhood until puberty is dominated by anaerobes due to low glycogen content, a decrease in Lactobacilli and other acrid-producing microbes and a more alkaline pH (30). This increases their susceptibility to genital infections (e.g., vulvovaginitis) by a multifariousness of aerobic and anaerobic pathogens including Due south. pyrogens, N. gonorrhea, E. coli, E. faecalis, C. vaginalis, Mycoplasmas, Diphtheroids, Bacteroides, S. epidermidis, C. albicans etc. (31–35). Fortunately, due to lack of exposure to sexual intercourse (coitus), the incidence of genital tract infections is low in children except in cases of child sexual corruption (36–38). At puberty, under the influence of rising estrogen levels, the vaginal epithelium thickens and stratifies, intracellular glycogen levels increase and undergo cyclical changes, cervicovaginal secretions are produced, and proliferation of lactic acid-producing lactobacilli commence (thirty, 39). The increasing production of lactic acid past Lactobacillus suggests there is a fermentable substrate present in the vagina. Glycogen is identified as the suitable carbohydrate substrate every bit an clan between loftier acrid secretion and presence of glycogen in the vagina was demonstrated over 80 years agone (xl). Vaginal glycogen is degraded by host α-amylase into maltose, maltotriose and α-dextrins, which are then converted to lactic acid by Lactobacilli (21, 39, 41–43). Elevated estrogen and glycogen levels promote increased thickness of the stratified squamous epithelium and protective mucus layer of the vagina (44). Lactic acid at physiological concentration (1% w/5, ~110 mM) (25, 26, 45) reduces the vaginal pH, which encourages the proliferation of Lactobacilli and inhibits the growth of the anaerobes and viruses capable of causing infection (26, 39, 45–49). It too exhibits some immunomodulatory furnishings on cervicovaginal epithelial cells and other cell types. It stimulates an anti-inflammatory country through the increased production of IL-ane receptor antagonist (IL-1RA) from cervicovaginal epithelial cells and inhibits the activation of nuclear factor- κB (NF-κB) in peripheral blood mononuclear cells and monocytes-macrophages (l, 51), which promotes the transcription of pro-inflammatory target genes. In improver, information technology inhibits the Toll-like receptor (TLR)-induced production of inflammatory mediators from cervicovaginal epithelial cells. Both D- and L-lactic acid showroom these anti-inflammatory furnishings that are potentiated by depression pH < 3.86 (22, 49, 52). The homeostatic vaginal environs created by lactobacillus-dominant microbiota is temporarily altered during catamenia when in that location is a reject in estrogen and glycogen levels, and neutralization of the acidic pH promoting the growth of pathogenic bacteria such every bit Gardnerella, Prevotella, and Atopobium (53). Afterward menstruation, the normal pH and Lactobacillus say-so are re-established as estrogen levels begin to ascent once again (54).
The vaginal microbiota in normal pregnancy is predominated by Lactobacilli species and is more stable than that in non-significant state (55, 56). This tin be explained by the college level of estrogen during pregnancy resulting in increased vaginal glycogen degradation, which enhances the proliferation of lactobacilli-dominated vaginal microbiota. Equally discussed subsequently in this article, oestriol, which is one of three major endogenous estrogens produced in significantly larger amounts by the placenta during pregnancy (57), was as strong equally 17β-oestradiol (the most mutual grade of estrogen in non-pregnant premenopausal women) in stimulating vaginal glycogen deposition (58).
Following menopause, Lactobacilli dominance decreases secondary to diminished estrogen levels (59, 60) and abeyance or decline of glycogen production (61). Loss of Lactobacilli increases vaginal pH to a more alkaline environment, providing a conducive habitat for colonization past anaerobes (possibly of fecal origin) and other pathogens (30, 62). For instance, post-menopausal women showed lower free genital fluid glycogen levels (41), and harbored more of G. vaginalis, Bacteroides, Peptostreptococcus, Streptococcus, and Prevotella (63) compared to premenopausal women who had higher gratuitous glycogen (41) and preponderance of L. crispatus and L. iners (64). Topical or systemic hormone replacement therapy and probiotics restore vaginal Lactobacillus authorisation and homeostasis with increased acerbity (65), and improves vaginal symptoms in mail service-menopausal women (44). In some cases, there is a decrease in the prevalence of anaerobic gram-negative rods and vaginal pH while the aerobic isolates including Lactobacilli remain fairly abiding (65).
Though progesterone lonely and in combination with estrogen greatly increases the glycogen concentration in the vaginal epithelium in squirrel monkeys (66), information technology does non appear to have the same outcome in humans (58). For instance, progestin-only contraceptive drugs such equally Depomedroxyprogesterone acetate (Depo-Provera) tin can produce a systemic hypoestrogenic state associated with slight thinning of the glycogen vaginal epithelial layer and reduced Lactobacillus colonization compromising the vaginal barrier against infection (67, 68). In fact, a pregnant negative correlation between free vaginal glycogen and progesterone level was recently reported in genital fluid samples of premenopausal women (42).
Also, the estrogenization and the subsequent production of glycogen past the vaginal epithelium accept been shown to promote infection by Candida albicans (69). Glycogen was suggested to exist a suitable substrate for C. albicans. This was particularly seen in post-menopausal women on systemic or vaginal estrogen therapy, whereas in a more contempo written report with premenopausal women, vulvovaginal candidiasis was observed in women with low vaginal α-amylase action (43). At that place is a demand to investigate the relationship betwixt α-amylase activeness and Lactobacillus authorisation in post-menopausal women on and without estrogen therapy.
Pathogenesis of Stress-Induced Vaginal Dysbiosis
The effect of stress on the incidence of BV may be mediated by stress-related dysregulation of immune role rather than behavioral changes associated with stress (20). An optimal allowed response is required to foreclose proliferation of BV-associated anaerobes. A sub-optimal response maybe secondary to genetic polymorphism increases the take a chance of infection (70), and may allow more ascending genital tract infection (71). Stress enhances the progression of infection (including BV) and its pathophysiologic consequences (72, 73).
Perceived psychosocial stress is significantly and independently related to incidence and prevalence of BV in pregnant (74) and not-significant women (20). Some stress stimuli that tin increase the run a risk of BV independent of individual behaviors include challenging life weather condition such depression income, poor housing, and dangerous neighborhood conditions, poor diet and interpersonal conflicts (73, 75). These studies assessed the level of stress exposure using the Cohen Perceived Stress Scale (20, 74, 75). Chronic psychosocial stress is also an established hazard factor for preterm delivery independent of other biomedical and behavioral risk factors (2). Stress regardless of pregnancy status tin range from acute and severe (due east.g., trauma), moderate (east.thou., alter in life events), and chronic (e.g., standing for long hours as part of daily occupation). Withal, during pregnancy, both short- and long-term stress exposure can atomic number 82 to agin pregnancy and delivery outcome (five, 76–78), though some studies with in vitro fertilization (IVF) patients argue otherwise showing only minimal clan betwixt prenatal stress and adverse pregnancy effect (79).
Stress-induced cortisol binds to glucocorticoid receptors expressed on a range of immune cells and alters NF-κB activities, which regulates the activity of inflammatory mediators such as cytokines (IL-1β, IL-6, TNF-α, IFNγ) and chemokines (IL-eight, CCL5). Glucocorticoids too facilitates immunosuppression past inhibiting proliferation, migration and cytotoxicity of lymphocytes and leukocytes, and secretion of IL-2 and IFN-γ (73). On the other hand, stress-induced epinephrine and norepinephrine bind to adrenergic receptors, activate army camp and stimulate the transcription of genes encoding for a variety of inflammatory mediators (9, eighty). Higher stress scores correlate with college levels of IL-half dozen and TNF-α; and with low levels of the anti-inflammatory cytokine IL-10 (5). Furthermore, stress hormones increase vulnerability to infections that are primarily prevented by innate and adaptive immune responses by differentially regulating monocyte, macrophage, Th1/Th2 cells and cytokine expression patterns (81–84). These cortisol and catecholamines-mediated changes in gene expression dysregulate immune role (nine). Nonetheless, with previous ascertainment of stress-related alterations in production of inflammatory mediators that are not associated with serum cortisol levels (85), the bear upon of stress on immune office may not exist express to cortisol-mediated effects alone (20).
A stress-induced vaginal dysbiosis with disrupted vaginal mucosal and immune response-related proteins (eastward.g., lactoferrin), reduction in neutrophil bactericidal potency and reduced abundance of commensal Lactobacillus take been demonstrated in a mouse model (86). Reduced Lactobacillus was associated with overgrowth of other microbes (86), most likely anaerobic and facultative bacteria. Exposure to stress produces a meaning decrease in the abundance of vaginal Lactobacillus species and tin can amplify the severity and consequences of vaginal infection (27). This occurs through the release of CRH, which stimulates sympathetic nerve terminals and the SAM axis to release norepinephrine. Norepinephrine can enter the vagina from the bloodstream and is locally secreted by vaginal epithelial cells to which information technology binds (80). Though the vaginal epithelial cells in this in vitro experiment were exposed to a high range of norepinephrine concentrations (ane-10 μM), this may non differ significantly from the in vivo environment. This is supported past evidence of abundant supply of norepinephrine from nerve terminals innervating the cervicovaginal mucosa in humans and animals (87–91) and vaginal epithelial cells could potentially collaborate with norepinephrine in the circulation every bit well (fourscore). In addition, local production of norepinephrine by epithelial cells in other sites such as the cornea has been demonstrated (92). The combined effect of a depleted vaginal Lactobacillus potency, which leads to decreased lactic acid and H2Oii production and increased alkalinity and proliferation of pathogenic bacteria; and increased norepinephrine secretion effect in a heightened pro-inflammatory response with increased production of cytokines and chemokines [(27); Figure 1). In essence, high norepinephrine levels induced by severe stress, potentiates the pro-inflammatory response of vaginal epithelial cells possibly in an autocrine fashion in the presence of macerated Lactobacilli and low pH (80).
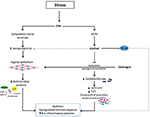
Figure 1. Stress-related reduction in vaginal Lactobacillus dominance (dysbiosis) and dysregulated immune response. Exposure to psychosocial stress induces the release of cortisol and norepinephrine via the hypothalamic-pituitary-adrenal and SAM axes respectively. Cortisol inhibits the estrogen-related vaginal epithelial maturation and glycogen accumulation. Levels of vaginal free glycogen and Lactobacilli are reduced leading to decreased lactic acrid and hydrogen peroxide (H2O2) synthesis and pH. Consequently, a dysbiotic environment conducive for the proliferation of bacterial vaginosis-associated anaerobic bacteria such as Gardnerella, Prevotella, Mobiluncus, Atopobium, Megasphera, and sexually transmitted infections such Neisseria gonorrhea, Chlamydia trachomatis, homo immunodeficiency virus is created. Cortisol too affects immune response past altering the nuclear cistron-κB (NF-κB) signal transduction pathway, which regulates inflammatory factor expression. These furnishings are exacerbated by the concurrent release of norepinephrine, which binds to vaginal epithelial cells and potentiates the pro-inflammatory response via a reduction in the release of antimicrobial proteins including mucins, immunoglobulins (secretory Ig A and IgG), β-defensins, secretory leucocyte protease inhibitor (SLPI), and neutrophil gelatinase-associated lipocalin (NGAL). The overall event is a dysbiotic vaginal ecosystem with a sub-optimal allowed response, which encourages upper genital tract infection with deleterious gynecological and obstetric sequelae.
Information technology also noteworthy that the incidence and prevalence of BV is influenced by other factors including frequent unprotected sexual activity with new or multiple partners (93–96), smoking, alcohol and drug utilize (73, 96–99), contraceptive practice (97, 98, 100), vaginal douching (95, 97, 101), period (102, 103), pregnancy (96, 104), low educational and socioeconomic status, and black race (73, 95–98, 100, 105). Nevertheless, after adjusting for all of these associated factors that usually business relationship for just minimal proportion of the variation in BV, the relationship between psychosocial stress and abnormal vaginal microbiota has been shown to be maintained in several studies (twenty, 74). The pathophysiology, risk factors and consequences of BV and other female genital tract infections take been reviewed extensively (22, 106), and more investigation is recommended due to the enigmatic nature of these conditions especially BV.
Cortisol Inhibits Vaginal Glycogen Deposition
The vaginal glycogen degradation action of estrogen was demonstrated over 50 years ago. Wrenn and colleagues in an endeavour to decide the estrogen content of biological fluids, observed a rapid increase in vaginal glycogen content later locally administering estrogen extracted from human and cow urine and blood samples to adolescent, ovariectomized rats (58). This was termed the "vaginal glycogen analysis for estrogen." Furthermore, they investigated the specificity of the assay by comparing the effect of 17β-oestradiol with progesterone, cortisol, deoxycorticosterone, testosterone and diethylstilboestrol (DES, synthetic not-steroidal estrogen), both individually and in combination with 17β-oestradiol. Of all these hormonal substances, which are equally applied intravaginally, only DES stimulated a vaginal glycogen response singly, which was significantly enhanced in combination with 17β-oestradiol. Interestingly, the hormones associated with stress i.east., cortisol (30 μg/0.01 ml) and deoxycorticosterone (40 μg/0.01 ml) exhibited moderate inhibition of the glycogen response when administered in combination with 17β-oestradiol, while progesterone and testosterone were ineffective in this regard. In improver, the vaginal glycogen action of the other ii naturally occurring forms of estrogen was tested. It was observed that oestrone which is ordinarily found in post-menopausal women was but about 10% every bit active as 17β-oestradiol, whereas oestriol (common during pregnancy) was as potent as 17β-oestradiol (58). This gives more acceptance to the more stable Lactobacilli dominated vaginal microbiota rich in glycogen and lactic acrid content observed in good for you pregnant women. Still, because this experiments were conducted in rats with relatively high concentrations of cortisol (8.iii × 106 nmol/50) compared to circulating cortisol levels under maximum stress such as major surgery (726 and 1297 nmol/l) (107, 108) or intravenous administration of 50 mg hydrocortisone (2450 nmol/l) (109) in humans, more empirical prove from human experiments is required. Perhaps the local (cervicovaginal) concentrations of cortisol as demonstrated by Wrenn et al. (58) is more crucial in the glycogen response than the influence of circulating cortisol as reported in humans. More so, it could exist that such repressive issue on vaginal glycogen deposition in humans could only be attained via a combined local action of cortisol and norepinephrine that mediate the physiologic stress response. This is withal to be demonstrated in human specimens.
The above evidence could be one mechanism through which frequent use of corticosteroids alters the equilibrium of the vaginal microbiome apart from dysregulated immune response. Cortisol repressed the estrogen-related maturation of vaginal epithelial cells and glycogen accumulation. If this happens in humans, the breakdown of glycogen to smaller polymers by α-amylase is reduced leading to low lactic acid production and ultimately loss of Lactobacillus dominance. This recipe creates a conducive, less acidic ecosystem for infection past strict and facultative anaerobic bacteria, viruses, fungi, and protozoa (27). Because the maturation of the vaginal epithelium is impaired, there is inadequate secretion of mucins and other antimicrobial proteins such every bit secretory leukocyte protease inhibitor (SLPI), neutrophil gelatinase-associated lipocalin (NGAL), and β-defensins. In addition, cortisol as well alters NF-κB activity and dysregulates the expression of pro-inflammatory cytokines and chemokines (9). The combined effect is a dysregulated/ineffective inflammatory response confronting the pathogens, and an uncontrolled and perhaps ascending genital tract infection, which can be deleterious especially during pregnancy.
Since stress is almost inevitable, these deportment of cortisol take a propensity to disrupt the equilibrium of the vaginal microbiome and its capacity to combat infectious agents. Excessive exposure to psychosocial stress is independently associated with increased prevalence of BV (xx, 73–75), which is the most common vaginal condition in women of reproductive age. BV is a quintessential dysbiotic condition characterized by overgrowth of anaerobic gram-negative and gram-variable bacteria secondary to a decrease in the protective Lactobacillus species. During pregnancy, BV has been linked to stress and it increases the chance of preterm labor by ~3-fold (110). Infections such as BV tin atomic number 82 to ascending intrauterine infection that stimulates immune responses with release of inflammatory mediators, uterotonins (Prostaglandins, PGs) and extracellular matrix degrading enzymes (e.g., matrix metalloproteinases, MMPs). These inflammatory cytokines, chemokines, PGs, and MMPs stimulate the pathway to premature rupture of membranes and preterm birth i.e., uterine contraction, cervical ripening, and membrane activation, via a positive feedback loop (22, 28, 106, 110).
While studies on stress and vaginal dysbiosis are often done without measurement of cortisol levels (xx, 74, 75, 111); and others did not detect a clear pattern of association between perceived stress (resulting from change in relationships, sickness/injury, finances, piece of work pressure or routines, unpleasant events, and relationship pressure) (111) or stress hormones and BV (112); the disruptive office of cortisol on the vaginal microbiota and immunity has been highlighted past the experiments of Wrenn et al. (58), albeit in animate being models studied with significantly college cortisol concentrations. Too, the correlation of cortisol levels with the prevalence of BV and other genitourinary tract infections in humans has been reported (113). In fact, a meaning increase in cortisol across the 2nd and third trimesters was observed in patients with BV, an association hypothesized to exist restricted to a local response to cortisol (113). Elevated cortisol levels could correlate with reduced vaginal glycogen content, loss of Lactobacillus dominance, decreased acidity, dysbiosis, and increase production of pro-inflammatory mediators. More then, information technology is plausible that increased norepinephrine activity, which in concurrence with cortisol is involved in the "fight-or-flight" response and has been shown to potentiate the pro-inflammatory response of vaginal epithelial cells, amplifies these actions of cortisol (Figure 1). Furthermore, since vaginal glycogen deposition is not required for immediate survival during exposure to stressful stimuli and subsequent "fight-or-flight" response, cortisol'southward action appears to be an adaptive mechanism to preserve free energy sources for organs such as the brain, cardiac and skeletal muscles albeit with unpleasant reproductive consequences when prolonged. A more than comprehensive investigation to elucidate the mechanisms of cortisol's activeness on the human vaginal microflora and the consequent gynecological and obstetric health implications is required.
Stress-Induced Contradistinct Maternal Vaginal and Offspring Gut Microbiota
Exposure to chronic psychosocial stress during pregnancy may amplify the physiologic pregnancy-induced immunosuppression (114). This can increment the gamble of dysbiosis, recurrent genitourinary infection and eventual loss of the beneficial lactobacillus-dominated vaginal microbiota equally seen in BV and candidiasis (115, 116). Since the initial neonatal gut microbial colonization is dependent on vertical transmission from the maternal vaginal microbiota during parturition, acquisition of a dysbiotic lactobacillus-depleted microbiota predisposes the babe to altered alimentary canal maturation, impaired extraction of free energy and macromolecules essential for normal growth and evolution, and dysfunctional immune organisation. This eventually increases the infant'due south risk of disturbed energy metabolism, obesity, insulin resistance, and diabetes mellitus. Other atmospheric condition include diarrheal illness, food allergy, atopic diseases, and inflammatory bowel disease, and irritable bowel syndrome (117, 118). Also, the risk of long-term neurodevelopmental disorders due to reprogramming of the developing brain has been reported (86, 114, 119). These findings are supported by human studies and experiments with animal models. For instance, infants of mothers with high prenatal stress (reported and due to high cortisol levels) had significantly lower relative abundance of lactic acid-producing bacteria including Lactobacillus, Lactoccus, Aerococcus, and Bifidobacteria; and higher relative abundance of Proteobacteria including the pathogenic Due east. coli, Serratia, and Enterobacter. This dysbiotic microbiota is potentially associated with an increased level of lipopolysaccharide (LPS)-induced inflammation and more maternally-reported infant gastrointestinal symptoms and allergic reactions (118).
Furthermore, female rhesus monkeys exposed to moderate stress (acoustic startle) for vi weeks in after gestation showed activated HPA axis and increased cortisol levels (above those constitute in normal pregnancy), that was sufficient to cause contradistinct intestinal microbiota characterized past decreased protective Lactobacilli and Bifidobacteria in their offsprings (120). Reduced concentrations of Lactobacilli and total and Gram-negative aerobes and facultative anaerobes accept also been observed in the small intestine of pups whose mothers were injected with cortisone before nascence (121, 122). These creature studies have provided ample evidence that the relationship between prenatal psychosocial stress and offspring programming is mediated, at least in part, past elevated cortisol levels (119). The pathophysiology of the effect of prenatal stress on maternal gut/vaginal and fetal gut microbial limerick, and the development of insulin resistance, increased adiposity and metabolic syndrome afterward in life in humans is beyond the scope of this review but requires further investigation.
Preventive and Treatment Strategies for Stress-Induced Vaginal Infection and Inflammation
Optimal preventive/treatment approach to counteract the outcome of stress on vthe aginal microenvironment should aim to alleviate or eliminate the stress stimuli and the associated infectious and inflammation agent(south). Though stress is almost inevitable, some life modifications tin can help an individual cope with stressors amend. These adaptive measures include regular physical exercise, healthy eating, having adequate sleep and fugitive unhealthy habits similar smoking, alcohol, and drug abuse. In addition to eliminating stressors and lifestyle modifications, accordingly administered antibiotics or antifungal agents can eliminate bacterial and fungal infections while inflammation tin can exist targeted pharmacologically using anti-inflammatory agents. This is an important therapeutic goal because inflammation can persist even after the stress stimuli or infectious agents take been eliminated (22). Antibiotic handling may also modify the vaginal bacterial composition with potential colonization by opportunistic pathogenic agents (123).
Another potential approach to restoring and/or maintaining vaginal homeostasis that avoids the agin effect of antibiotics on beneficial microbiota (124) is the employ of probiotics e.g., Lactobacillus species (L. crispatus), and prebiotics such as D-lactic acid, glycogen, oligosaccharide etc. that stimulate probiotic bacterial growth (22, 124–131). Recently, in a study including 6 women, 5 of whom were significant, vaginal, and oral lactoferrin assistants improved vaginal microbiota Lactobacillus dominance and prevented refractory BV, cervical inflammation, and preterm delivery (132). This has previously been attempted with similar results past the same group (133) and others (134); and gives acceptance to the observed altered vaginal microbiota (with decreased Lactobacilli and lactoferrin levels) when mice were exposed to mild prenatal stress (86). A combination of probiotics and prebiotics could provide clinically useful offshoot to pharmacological and other strategies for treating/preventing vaginal infections and women'south health in general (129, 131).
Some anti-inflammatory agents that tin can block LPS-driven production of cytokines (due east.chiliad., TNF-α, IL-half-dozen and IL-1β) every bit observed in stress-induced vaginal colonization by anaerobic bacteria include phosphodiesterase (PDE) inhibitors, MAP kinase inhibitors, TNF biologics and NF-κB inhibitors (135). Some of these agents have also been applied in the prevention of LPS-stimulated preterm birth and fetal death (136, 137). With farther exploration of the clinical utility, safety and toxicity of these anti-inflammatory agents, various stages of the stress/infection-associated inflammatory process can be targeted. However, because NF-κB is the "principal regulator" of inflammatory gene expression, there is the crucial concern that inhibiting the activation of NF-κB and/or other pro-inflammatory mediators could forestall optimal activation of host innate and adaptive immune responses and increase the take a chance of infection. This could be deleterious particularly in an immunocompromised country such every bit pregnancy (135, 138).
Main Findings and Future Management
The potential action of the stress hormone cortisol in the maintenance of vaginal health has been highlighted. Stress stimuli are ubiquitous and women practice non enjoy any exemptions. The physiologic "fight-or-flight" response may be deleterious to their lower genital tract microbiome if the stress stimuli persist for longer than necessary. Persistent exposure to psychosocial stress and stimulation of the HPA and SAM axes, with a corresponding increase in cortisol and norepinephrine levels are associated with dysbiosis and increased susceptibility to several infections including genitourinary tract infections. Though this could exist solely due to a dysregulated allowed response, a cortisol-induced inhibition of vaginal glycogen deposition may be involved specially in the case of vaginal infection. The estrogen-related increased vaginal glycogen deposition and epithelial maturation are requirements for the maintenance of vaginal eubiosis. The ability of cortisol to disrupt this process as shown in beast models is important in the pathogenesis of vaginal dysbiosis and subsequent development of infection and inflammation. If proven in humans, this phenomenon may be more than crucial in pregnancy where a good for you Lactobacillus-dominated vaginal microbiome is sacrosanct, and at that place is local production of more CRH from gestational tissues including the decidua, fetal membranes (chorioamnion), and placenta. Considering the current evidence of the repressive role of cortisol on vaginal glycogen deposition involve experiments in animals with high concentrations of cortisol compared to that observed during severe stress in humans, further piece of work especially in humans is needed to elucidate the pathophysiologic association between cortisol levels and vaginal microbiomial architecture and part.
Author Contributions
EA conceived the enquiry idea and conducted the literature search. EA and DA contributed to writing the manuscript. Both authors revised and approved the submission of the final manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could exist construed equally a potential disharmonize of interest.
Abbreviations
ACTH, adrenocorticotropic hormone; BV, bacterial vaginosis; cAMP, circadian adenosine monophosphate; CCL5, Chemokine (C-C motif) ligand 5; CRH, corticotropin-releasing hormone; DES, diethylstilboestrol; HPA, hypothalamic-pituitary-adrenal; IFNγ, interferon gamma; IL, interleukin; IL-1RA, interleukin-1 receptor antagonist; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; NGAL, neutrophil gelatinase-associated lipocalin; PVN, paraventricular nucleus; SAM, sympathetic-adrenal-medullary; SLPI, secretory leucocyte protease inhibitor; TLR, price-like receptors; TNF-α, tumor necrosis cistron alpha.
References
1. Wadhwa PD, Culhane JF, Rauh Five, Barve SS. Stress and preterm nascency: neuroendocrine, allowed/inflammatory, and vascular mechanisms. Matern Kid Health J. (2001) five:119–25. doi: x.1023/A:1011353216619
PubMed Abstract | CrossRef Full Text | Google Scholar
two. Wadhwa PD, Culhane JF, Rauh V, Barve SS, Hogan V, Sandman CA, et al. Stress, infection and preterm birth: a biobehavioural perspective. Paediatr Perinat Epidemiol. (2001) 15(Suppl. 2):17–29. doi: ten.1046/j.1365-3016.2001.00005.x
PubMed Abstract | CrossRef Total Text | Google Scholar
3. Chrousos GP. Regulation and dysregulation of the hypothalamic-pituitary-adrenal axis. The corticotropin-releasing hormone perspective. Endocrinol Metab Clin Northward Am. (1992) 21:833–58. doi: 10.1016/S0889-8529(eighteen)30191-9
PubMed Abstract | CrossRef Full Text | Google Scholar
4. Orr ST, James SA, Casper R. Psychosocial stressors and low birth weight: development of a questionnaire. J Dev Behav Pediatr. (1992) 13:343–7. doi: 10.1097/00004703-199210010-00005
PubMed Abstract | CrossRef Total Text | Google Scholar
five. Coussons-Read ME, Okun ML, Schmitt MP, Giese Due south. Prenatal stress alters cytokine levels in a manner that may endanger homo pregnancy. Psychosom Med. (2005) 67:625–31. doi: 10.1097/01.psy.0000170331.74960.ad
PubMed Abstruse | CrossRef Full Text | Google Scholar
6. Sapolsky RM, Romero LM, Munck AU. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative deportment. Endocr Rev. (2000) 21:55–89. doi: 10.1210/er.21.1.55
PubMed Abstract | CrossRef Full Text | Google Scholar
7. Munck A, Náray-Fejes-Tóth A. The ups and downs of glucocorticoid physiology Permissive and suppressive furnishings revisited. Mol Jail cell Endocrinol. (1992) 90:C1–4. doi: ten.1016/0303-7207(92)90091-J
PubMed Abstract | CrossRef Full Text | Google Scholar
8. Chrousos GP, Kino T. Glucocorticoid action networks and circuitous psychiatric and/or somatic disorders. Stress (2007) ten:213–9. doi: 10.1080/10253890701292119
CrossRef Full Text | Google Scholar
eleven. Shrayyef MZ, Gerich JE. Normal glucose homeostasis. In: Poretsky L, editor. Principles of Diabetes Mellitus. 2nd ed. Boston, MA: Springer (2010) p. 19–35.
Google Scholar
12. Kamba A, Daimon K, Murakami H, Otaka H, Matsuki Thousand, Sato Due east, et al. Association between higher serum cortisol levels and decreased insulin secretion in a general population. PLOS ONE (2016) 11:e0166077. doi: 10.1371/journal.pone.0166077
PubMed Abstract | CrossRef Total Text | Google Scholar
thirteen. Chung S, Son GH, Kim K. Circadian rhythm of adrenal glucocorticoid: Its regulation and clinical implications. Biochim Biophys Acta (2011) 1812:581–91. doi: 10.1016/j.bbadis.2011.02.003
PubMed Abstract | CrossRef Full Text | Google Scholar
xiv. Knight RP Jr, Kornfeld, DS, Glaser, GH, Bondy PK. Effects of intravenous hydrocortisone on electrolytes of serum and urine in man. J Clin Endocrinol Metab. (1955) fifteen:176–81. doi: 10.1210/jcem-15-2-176
PubMed Abstract | CrossRef Full Text | Google Scholar
15. Sandle GI, Keir MJ, Record CO. The effect of hydrocortisone on the transport of water, sodium, and glucose in the jejunum. Scand J Gastroenterol. (1981) 16:667–71. doi: 10.3109/00365528109182028
PubMed Abstract | CrossRef Full Text | Google Scholar
16. Ackermann S, Hartmann F, Papassotiropoulos A, Quervain DJ-Fd, Rasch B. Associations between basal cortisol levels and memory retrieval in healthy immature individuals. J Cogn Neurosci. (2013) 25:1896–907. doi: 10.1162/jocn_a_00440
PubMed Abstract | CrossRef Full Text | Google Scholar
18. Stratakis CA, Chrousos GP. Neuroendocrinology and pathophysiology of the stress system. Ann N Y Acad Sci. (1995) 771:1–18.
PubMed Abstract | Google Scholar
nineteen. Debono M, Ghobadi C, Rostami-Hodjegan A, Huatan H, Campbell MJ, Newell-Price J, et al. Modified-Release Hydrocortisone to provide circadian cortisol profiles. J Clin Endocrinol Metab. (2009) 94:1548–54. doi: 10.1210/jc.2008-2380
PubMed Abstract | CrossRef Full Text | Google Scholar
20. Nansel TR, Riggs MA, Klebanoff M. The association of psychosocial stress and bacterial vaginosis in a longitudinal accomplice. Am J Obstet Gynecol. (2006) 194:381–vi. doi: 10.1016/j.ajog.2005.07.047
PubMed Abstract | CrossRef Full Text | Google Scholar
21. Mirmonsef P, Hotton AL, Gilbert D, Burgad D, Landay A, Weber KM, et al. Free glycogen in vaginal fluids is associated with Lactobacillus colonization and low vaginal pH. PLoS ONE (2014) 9:e102467. doi: x.1371/periodical.pone.0102467
PubMed Abstract | CrossRef Total Text | Google Scholar
23. Petrova MI, Lievens E, Malik S, Imholz N, Lebeer S. Lactobacillus species as biomarkers and agents that can promote various aspects of vaginal health. Front Physiol. (2015) vi:81. doi: 10.3389/fphys.2015.00081
PubMed Abstract | CrossRef Full Text | Google Scholar
24. Ravel J, Gajer P, Abdo Z, Schneider GM, Koenig SS, McCulle SL, et al. Vaginal microbiome of reproductive-age women. Proc Natl Acad Sci United states of america. (2011) 108(Suppl. 1):4680–7. doi: ten.1073/pnas.1002611107
PubMed Abstract | CrossRef Full Text | Google Scholar
25. Aldunate M, Srbinovski D, Hearps Air-conditioning, Latham CF, Ramsland PA, Gugasyan R, et al. Antimicrobial and immune modulatory effects of lactic acid and curt chain fatty acids produced past vaginal microbiota associated with eubiosis and bacterial vaginosis. Front Physiol. (2015) 6:164. doi: 10.3389/fphys.2015.00164
PubMed Abstract | CrossRef Full Text | Google Scholar
28. Witkin Southward. The vaginal microbiome, vaginal anti-microbial defence mechanisms and the clinical challenge of reducing infection-related preterm nascence. BJOG (2015) 122:213–8. doi: 10.1111/1471-0528.13115
CrossRef Full Text | Google Scholar
31. Dasari S, Anandan SK, Rajendra West, Valluru 50. Role of microbial flora in female genital tract: a comprehensive review. Asian Pacific J Trop Dis. (2016) half-dozen:909–17. doi: x.1016/S2222-1808(16)61155-half dozen
CrossRef Full Text | Google Scholar
32. Hammerschlag MR, Alpert S, Onderdonk AB, Thurston P, Drude Eastward, McCormack WM, et al. Anaerobic microflora of the vagina in children. Am J Obstet Gynecol. (1978) 131:853–6. doi: 10.1016/S0002-9378(16)33130-1
PubMed Abstract | CrossRef Full Text | Google Scholar
33. Hammerschlag MR, Alpert Southward, Rosner I, Thurston P, Semine D, McComb D, et al. Microbiology of the vagina in children: normal and potentially pathogenic organisms. Pediatrics (1978) 62:57–62.
PubMed Abstract | Google Scholar
34. Gerstner GJ, Grunberger W, Boschitsch East, Rotter Yard. Vaginal organisms in prepubertal children with and without vulvovaginitis. A vaginoscopic study. Arch Gynecol. (1982) 231:247–52. doi: x.1007/BF02110125
PubMed Abstract | CrossRef Full Text | Google Scholar
35. Bumbuliene Ž, Venclavičiute K, Ramašauskaite D, Arlauskiene A, Bumbul E, Drasutiene K. Microbiological findings of vulvovaginitis in prepubertal girls. Postgraduate Med J. (2014) xc:8–12. doi: x.1136/postgradmedj-2013-131959
PubMed Abstract | CrossRef Full Text | Google Scholar
36. Jaquiery A, Stylianopoulos A, Hogg G, Grover S. Vulvovaginitis: clinical features, aetiology, and microbiology of the genital tract. Arch Dis Child (1999) 81:64–seven.
PubMed Abstruse | Google Scholar
39. Spear GT, French AL, Gilbert D, Zariffard MR, Mirmonsef P, Sullivan TH, et al. Human α-amylase present in lower genital tract mucosal fluid processes glycogen to back up vaginal colonization past Lactobacillus. J Infect Dis. (2014): 210:1019–28. doi: 10.1093/infdis/jiu231
PubMed Abstract | CrossRef Full Text | Google Scholar
40. Robert C. The conversion of the glycogen of the vagina into lactic acid. J Pathol Bacteriol. (1934) 39:213–9. doi: ten.1002/path.1700390118
CrossRef Full Text | Google Scholar
41. Mirmonsef P, Modur Due south, Burgad D, Gilbert D, Golub ET, French AL, et al. An exploratory comparison of vaginal glycogen and Lactobacillus levels in pre- and mail service-menopausal women. Menopause (2015) 22:702–9. doi: ten.1097/GME.0000000000000397
CrossRef Full Text | Google Scholar
42. Mirmonsef P, Hotton AL, Gilbert D, Gioia CJ, Maric D, Hope TJ, et al. Glycogen levels in undiluted genital fluid and their relationship to vaginal pH, estrogen, and progesterone. PLoS 1 (2016) 11:e0153553. doi: ten.1371/journal.pone.0153553
PubMed Abstract | CrossRef Total Text | Google Scholar
43. Nasioudis D, Beghini J, Bongiovanni AM, Giraldo PC, Linhares IM, Witkin SS. α-amylase in vaginal fluid:association with conditions favorable to dominance of Lactobacillus. Reprod Sci. (2015) 22:1393–viii. doi: 10.1177/1933719115581000
PubMed Abstract | CrossRef Full Text | Google Scholar
45. Aldunate M, Tyssen D, Johnson A, Zakir T, Sonza South, Moench T, et al. Vaginal concentrations of lactic acid potently inactivate HIV. J Antimicrob Chemoth. (2013) 68:2015–25. doi: ten.1093/jac/dkt156
PubMed Abstract | CrossRef Total Text | Google Scholar
47. Tyssen D, Wang YY, Hayward JA, Agius PA, DeLong M, Aldunate 1000, et al. Anti-HIV-i activity of lactic acid in man cervicovaginal fluid. mSphere (2018) 3:e00055–18. doi: 10.1128/mSphere.00055-18
PubMed Abstract | CrossRef Full Text | Google Scholar
48. Aldunate Thousand, Tyssen D, Latham C, Ramsland P, Perlmutter P, Moench T, et al. Vaginal concentrations of lactic acid potently inactivate HIV-1 compared to curt concatenation fatty acids present during bacterial vaginosis. AIDS Res Hum Retrovirus. (2014) thirty:2015–25. doi: 10.1089/assist.2014.5499.abstruse
CrossRef Full Text | Google Scholar
49. Hearps AC, Tyssen D, Srbinovski D, Bayigga L, Diaz DJD, Aldunate M, et al. Vaginal lactic acrid elicits an anti-inflammatory response from human cervicovaginal epithelial cells and inhibits production of pro-inflammatory mediators associated with HIV acquisition. Mucosal Immunol. (2017) 10:1480. doi: 10.1038/mi.2017.27
PubMed Abstract | CrossRef Full Text | Google Scholar
50. Lee JM, Hwang KT, Jun WJ, Park CS, Lee MY. Antiinflammatory effect of lactic acid bacteria: inhibition of cyclooxygenase-ii by suppressing nuclear factor-kappaB in Raw264.7 macrophage cells. J Microbiol Biotechnol. (2008) xviii:1683–eight.
PubMed Abstract | Google Scholar
51. Ménard S, Candalh C, Bambou JC, Terpend K, Cerf-Bensussan Due north, Heyman M. Lactic acid leaner secrete metabolites retaining anti-inflammatory properties after abdominal ship. Gut (2004) 53:821–8. doi: 10.1136/gut.2003.026252
PubMed Abstruse | CrossRef Full Text | Google Scholar
52. Tachedjian G, Aldunate Yard, Bradshaw CS, Cone RA. The role of lactic acrid production past probiotic Lactobacillus species in vaginal health. Res Microbiol. (2017) 168:782–92. doi: 10.1016/j.resmic.2017.04.001
PubMed Abstract | CrossRef Full Text | Google Scholar
53. Lopes dos Santos Santiago One thousand, Tency I, Verstraelen H, Verhelst R, Trog Grand, Temmerman Yard, et al. Longitudinal qPCR report of the dynamics of L. crispatus, L. iners, A. vaginae, (Sialidase Positive) G. vaginalis, P. bivia in the Vagina. PLoS ONE (2012) 7:e45281. doi: x.1371/journal.pone.0045281
CrossRef Full Text | Google Scholar
54. Srinivasan S, Liu C, Mitchell CM, Fiedler TL, Thomas KK, Agnew KJ, et al. Temporal variability of human vaginal bacteria and relationship with bacterial vaginosis. PloS Ane (2010) 5:e10197. doi: 10.1371/journal.pone.0010197
PubMed Abstract | CrossRef Full Text | Google Scholar
55. Romero R, Hassan SS, Gajer P, Tarca AL, Fadrosh DW, Nikita L, et al. The composition and stability of the vaginal microbiota of normal pregnant women is different from that of non-significant women. Microbiome (2014) 2:4. doi: x.1186/2049-2618-2-4
CrossRef Full Text | Google Scholar
56. DiGiulio DB, Callahan BJ, McMurdie PJ, Costello EK, Lyell DJ, Robaczewska A, et al. Temporal and spatial variation of the man microbiota during pregnancy. Proc Natl Acad Sci USA. (2015) 112:11060–5. doi: 10.1073/pnas.1502875112
PubMed Abstract | CrossRef Full Text | Google Scholar
57. Mesiano Southward. Affiliate 11 - The endocrinology of human pregnancy and fetoplacental neuroendocrine development. In: Strauss JF Barbieri RL, editors. Yen & Jaffe's Reproductive Endocrinology. 6th ed. Philadelphia, PA: Due west.B. Saunders (2009) p. 249–81.
58. Wrenn TR, Forest JR, Bitman J, Brinsfield Thursday. Vaginal glycogen assay for oestrogen: specificity and application to blood and urine. J Reprod Fertil. (1968) 16:301–4. doi: 10.1530/jrf.0.0160301
PubMed Abstract | CrossRef Full Text | Google Scholar
59. Cauci S, Driussi S, De Santo D, Penacchioni P, Iannicelli T, Lanzafame P, et al. Prevalence of bacterial vaginosis and vaginal flora changes in peri- and postmenopausal women. J Clin Microbiol. (2002) 40:2147–52. doi: 10.1128/JCM.twoscore.six.2147-2152.2002
PubMed Abstract | CrossRef Full Text | Google Scholar
60. Gupta S, Kumar N, Singhal N, Kaur R, Manektala U. Vaginal microflora in postmenopausal women on hormone replacement therapy. Indian J Pathol Microbiol. (2006) 49:457–61.
PubMed Abstract | Google Scholar
61. Cruickshank R, Sharman A. The biology of the vagina in the human discipline. BJOG (1934) 41:208–26. doi: 10.1111/j.1471-0528.1934.tb08759.x
CrossRef Total Text | Google Scholar
63. Burton JP, Reid One thousand. Evaluation of the bacterial vaginal flora of 20 postmenopausal women by straight (Nugent score) and molecular (polymerase chain reaction and denaturing gradient gel electrophoresis) techniques. J Infect Dis. (2002) 186:1770–80. doi: 10.1086/345761
PubMed Abstract | CrossRef Full Text | Google Scholar
64. Brotman RM, Shardell Doctor, Gajer P, Fadrosh D, Chang K, Silvery MI. Association betwixt the vaginal microbiota, menopause status, and signs of vulvovaginal atrophy. Menopause (2014) 21:450–viii. doi: 10.1097/GME.0b013e3182a4690b
PubMed Abstract | CrossRef Full Text | Google Scholar
65. Ginkel PD, Soper DE, Bump RC, Dalton HP. Vaginal flora in postmenopausal women: the effect of estrogen replacement. Infect Dis Obstet Gynecol. (1993) i:94–seven. doi: 10.1155/S1064744993000225
PubMed Abstract | CrossRef Full Text | Google Scholar
66. Bo WJ. The effect of progesterone and progesterone-estrogen on the glycogen degradation in the vagina of the squirrel monkey. Am J Obstet Gynecol. (1970) 107:524–thirty. doi: 10.1016/S0002-9378(16)33936-9
PubMed Abstruse | CrossRef Total Text | Google Scholar
67. Miller Fifty, Patton DL, Meier A, Thwin SS, Hooton TM, Eschenbach DA. Depomedroxyprogesterone-induced hypoestrogenism and changes in vaginal flora and epithelium. Obstet Gynecol. (2000) 96:431–ix. doi: ten.1016/S0029-7844(00)00906-6
PubMed Abstract | CrossRef Full Text | Google Scholar
68. Eschenbach DA, Patton DL, Meier A, Thwin SS, Aura J, Stapleton A, et al. Effects of oral contraceptive pill utilise on vaginal flora and vaginal epithelium. Contraception (2000) 62:107–12. doi: 10.1016/S0010-7824(00)00155-4
PubMed Abstract | CrossRef Full Text | Google Scholar
70. Goepfert AR, Varner G, Ward K, Macpherson C, Klebanoff M, Goldenberg RL, et al. Differences in inflammatory cytokine and Price-like receptor genes and bacterial vaginosis in pregnancy. Am J Obstet Gynecol. (2005) 193:1478–85. doi: 10.1016/j.ajog.2005.03.053
PubMed Abstract | CrossRef Full Text | Google Scholar
72. Biondi One thousand, Zannino LG. Psychological stress, neuroimmunomodulation, and susceptibility to infectious diseases in animals and man: a review. Psychother Psychosom. (1997) 66:3–26. doi: 10.1159/000289101
PubMed Abstract | CrossRef Full Text | Google Scholar
74. Culhane JF, Rauh Five, McCollum KF, Hogan VK, Agnew M, Wadhwa PD. Maternal stress is associated with bacterial vaginosis in human pregnancy. Matern Kid Health J. (2001) 5:127–34. doi: x.1023/A:1011305300690
PubMed Abstract | CrossRef Full Text | Google Scholar
75. Culhane JF, Rauh Five, McCollum KF, Elo It, Hogan 5. Exposure to chronic stress and ethnic differences in rates of bacterial vaginosis among pregnant women. Am J Obstet Gynecol. (2002) 187:1272–six. doi: 10.1067/mob.2002.127311
PubMed Abstruse | CrossRef Full Text | Google Scholar
77. Austin MP, Leader Fifty. Maternal stress and obstetric and infant outcomes: epidemiological findings and neuroendocrine mechanisms. Aust Due north Z J Obstet Gynaecol. (2000) 40:331–7. doi: x.1111/j.1479-828X.2000.tb03344.10
PubMed Abstract | CrossRef Full Text | Google Scholar
78. Gennaro S, Fehder WP. Stress, immune role, and relationship to pregnancy effect. Nurs Clin N Am. (1996) 31:293–303.
PubMed Abstract | Google Scholar
79. Milad MP, Klock SC, Moses S, Chatterton R. Stress and anxiety do not issue in pregnancy wastage. Hum Reprod. (1998) thirteen:2296–300. doi: 10.1093/humrep/13.8.2296
CrossRef Total Text | Google Scholar
lxxx. Brosnahan AJ, Vulchanova L, Witta SR, Dai Y, Jones BJ, Brown DR. Norepinephrine potentiates proinflammatory responses of man vaginal epithelial cells. J Neuroimmunol. (2013) 259:8–sixteen. doi: ten.1016/j.jneuroim.2013.03.005
PubMed Abstruse | CrossRef Full Text | Google Scholar
81. Elenkov IJ, Chrousos GP. Stress hormones, Th1/Th2 patterns, pro/anti-inflammatory cytokines and susceptibility to disease. Trends Endocrinol Metab. (1999) 10:359–68.
PubMed Abstruse | Google Scholar
83. Elenkov IJ, Webster EL, Torpy DJ, Chrousos GP. Stress, corticotropin-releasing hormone, glucocorticoids, and the immune/inflammatory response: acute and chronic furnishings. Ann N Y Acad Sci. (1999) 876:i–xi; discussion:3.
PubMed Abstruse | Google Scholar
84. Chrousos GP. Stressors stress, and neuroendocrine integration of the adaptive response. The 1997 Hans Selye Memorial Lecture. Ann N Y Acad Sci. (1998) 851:311–35.
PubMed Abstract | Google Scholar
85. Song C, Kenis Yard, van Gastel A, Bosmans East, Lin A, de Jong R, et al. Influence of psychological stress on immune-inflammatory variables in normal humans. Function Two. Altered serum concentrations of natural anti-inflammatory agents and soluble membrane antigens of monocytes and T lymphocytes. Psychiatry Res. (1999) 85:293–303. doi: 10.1016/S0165-1781(99)00012-8
PubMed Abstract | CrossRef Full Text | Google Scholar
86. Jašarević E, Howerton CL, Howard CD, Bale TL. Alterations in the vaginal microbiome by maternal stress are associated with metabolic reprogramming of the offspring gut and brain. Endocrinology (2015) 156:3265–76. doi: 10.1210/en.2015-1177
PubMed Abstract | CrossRef Total Text | Google Scholar
87. Pauls R, Mutema 1000, Segal J, Silva WA, Kleeman S, Dryfhout V, et al. Original research—bones scientific discipline: a prospective study examining the anatomic distribution of nerve density in the man vagina. J Sex Med. (2006) 3:979–87. doi: x.1111/j.1743-6109.2006.00325.ten
CrossRef Total Text | Google Scholar
88. Hilliges Chiliad, Falconer C, Ekman-Ordeberg G, Johansson O. Innervation of the human vaginal mucosa as revealed by PGP 9.5 immunohistochemistry. Cells Tissues Organs (1995) 153:119–26. doi: 10.1159/000147722
PubMed Abstract | CrossRef Total Text | Google Scholar
89. Mónica Brauer M, Smith PG. Estrogen and female reproductive tract innervation: cellular and molecular mechanisms of autonomic neuroplasticity. Auton Neurosci. (2015) 187:i–17. doi: x.1016/j.autneu.2014.11.009
CrossRef Full Text | Google Scholar
90. Owman C, Rosengren Eastward, Sjöberg NO. Adrenergic innervation of the human female reproductive organs: a histochemical and chemical investigation. Obstet Gynecol. (1967) thirty:763–73.
PubMed Abstract | Google Scholar
92. Pullar CE, Zhao M, Song B, Pu J, Reid B, Ghoghawala S, et al. ß-adrenergic receptor agonists delay while antagonists accelerate epithelial wound healing: evidence of an endogenous adrenergic network inside the corneal epithelium. J Cell Physiol. (2007) 211:261–72. doi: 10.1002/jcp.20934
CrossRef Full Text | Google Scholar
93. Verstraelen H, Verhelst R, Vaneechoutte Thousand, Temmerman M. The epidemiology of bacterial vaginosis in relation to sexual behaviour. BMC Infect Dis. (2010) 10:81. doi: 10.1186/1471-2334-10-81
PubMed Abstract | CrossRef Total Text | Google Scholar
94. Nilsson U, Hellberg D, Shoubnikova M, Nilsson South, Mårdh PA. Sexual behavior take a chance factors associated with bacterial vaginosis and Chlamydia trachomatis Infection. Sex Transm Dis. (1997) 24:241–6. doi: 10.1097/00007435-199705000-00001
PubMed Abstract | CrossRef Full Text | Google Scholar
95. Allsworth JE, Peipert JF. Prevalence of bacterial vaginosis: 2001–2004 national health and nutrition examination survey information. Obstet Gynecol. (2007) 109:114–xx. doi: x.1097/01.AOG.0000247627.84791.91
PubMed Abstract | CrossRef Total Text | Google Scholar
96. Koumans EH, Sternberg Yard, Bruce C, McQuillan G, Kendrick J, Sutton Thou, et al. The prevalence of bacterial vaginosis in the Us, 2001–2004; associations with symptoms, sexual behaviors, and reproductive wellness. Sexual practice Transm Dis. (2007) 34:864–nine. doi: ten.1097/OLQ.0b013e318074e565
PubMed Abstract | CrossRef Full Text | Google Scholar
97. Cherpes TL, Hillier SL, Meyn LA, Busch JL, Krohn MA. A frail balance: take chances factors for conquering of bacterial vaginosis include sexual activity, absence of hydrogen peroxide-producing lactobacilli, black race, and positive herpes simplex virus type two serology. Sex Transm Dis. (2008) 35:78–83. doi: x.1097/OLQ.0b013e318156a5d0
PubMed Abstruse | CrossRef Full Text | Google Scholar
98. Ness RB, Hillier Southward, Richter HE, Soper DE, Stamm C, Bass DC, et al. Can known chance factors explicate racial differences in the occurrence of bacterial vaginosis? J Natl Med Assoc. (2003) 95:201–12.
PubMed Abstruse | Google Scholar
101. Luong ML, Libman M, Dahhou K, Chen MF, Kahn SR, Goulet L, et al. Vaginal douching, bacterial vaginosis, and spontaneous preterm nascence. J Obstet Gynaecol Can. (2010) 32:313–twenty. doi: x.1016/S1701-2163(16)34474-7
PubMed Abstract | CrossRef Total Text | Google Scholar
102. Morison L, Ekpo G, West B, Demba Due east, Mayaud P, Coleman R, et al. Bacterial vaginosis in relation to menstrual cycle menstrual protection method, and sexual intercourse in rural Gambian women. Sex Transm Infect. (2005) 81:242–seven. doi: x.1136/sti.2004.011684
PubMed Abstract | CrossRef Full Text | Google Scholar
103. Eschenbach DA, Thwin SS, Patton DL, Hooton TM, Stapleton AE, Agnew K, et al. Influence of the normal menstrual cycle on vaginal tissue, discharge, and microflora. Clin Infect Dis. (2000) 30:901–7. doi: x.1086/313818
PubMed Abstract | CrossRef Full Text | Google Scholar
105. Tolosa JE, Chaithongwongwatthana S, Daly S, Maw WW, Gaita H, Lumbiganon P, et al. The International infections in pregnancy report:variations in the prevalence of bacterial vaginosis and distribution of morphotypes in vaginal smears amongst significant women. Am J Obstet Gynecol. (2006) 195:1198–204. doi: 10.1016/j.ajog.2006.08.016
PubMed Abstract | CrossRef Full Text | Google Scholar
106. Amabebe E. Analysis of Cervicovaginal Fluid Metabolome and Microbiome in Relation to Preterm Nascency. Ph.D. thesis, White Rose eTheses Online: University of Sheffield (2016).
108. Plumpton FS, Besser GM. The adrenocortical response to surgery and insulin-induced hypoglycaemia in corticosteroid-treated and normal subjects. BJS (1969) 56:216–9. doi: ten.1002/bjs.1800560315
PubMed Abstruse | CrossRef Full Text | Google Scholar
109. Jung C, Greco S, Nguyen HHT, Ho JT, Lewis JG, Torpy DJ, et al. Plasma, salivary and urinary cortisol levels following physiological and stress doses of hydrocortisone in normal volunteers. BMC Endocrine Disord. (2014) 14:91. doi: 10.1186/1472-6823-14-91
PubMed Abstruse | CrossRef Full Text | Google Scholar
110. Shapiro GD, Fraser WD, Frasch MG, Séguin JR. Psychosocial stress in pregnancy and preterm birth: associations and mechanisms. J Perinat Med. (2013) 41:631–45. doi: 10.1515/jpm-2012-0295
PubMed Abstruse | CrossRef Full Text | Google Scholar
112. Harville EW, Savitz DA, Dole N, Thorp JM Jr, Herring AH. Psychological and biological markers of stress and bacterial vaginosis in meaning women. BJOG (2007) 114:216–23. doi: x.1111/j.1471-0528.2006.01209.x
PubMed Abstruse | CrossRef Full Text | Google Scholar
113. Ruiz RJ, Fullerton J, Brown CEL, Schoolfield J. Relationships of cortisol, perceived stress, genitourinary infections, and fetal fibronectin to gestational age at nascence. Biol Res Nurs. (2001) 3:39–48. doi: 10.1177/109980040100300106
PubMed Abstract | CrossRef Full Text | Google Scholar
114. Jašarević E, Rodgers AB, Bale TL. A novel role for maternal stress and microbial transmission in early life programming and neurodevelopment. Neurobiol Stress (2015) 1:81–viii. doi: 10.1016/j.ynstr.2014.10.005
PubMed Abstract | CrossRef Total Text | Google Scholar
115. Gupta K, Stapleton AE, Hooton TM, Roberts PL, Fennell CL, Stamm WE. Inverse association of H2O2-producing lactobacilli and vaginal Escherichia coli colonization in women with recurrent urinary tract infections. J Infect Dis. (1998) 178:446–50.
PubMed Abstract | Google Scholar
116. Ehrström SM, Kornfeld D, Thuresson J, Rylander E. Signs of chronic stress in women with recurrent candida vulvovaginitis. Am J Obstet Gynecol. (2005) 193:1376–81. doi: 10.1016/j.ajog.2005.03.068
PubMed Abstruse | CrossRef Full Text | Google Scholar
118. Zijlmans MAC, Korpela 1000, Riksen-Walraven JM, de Vos WM, de Weerth C. Maternal prenatal stress is associated with the infant intestinal microbiota. Psychoneuroendocrinology (2015) 53:233–45. doi: ten.1016/j.psyneuen.2015.01.006
PubMed Abstract | CrossRef Total Text | Google Scholar
119. Beijers R, Buitelaar JK, de Weerth C. Mechanisms underlying the furnishings of prenatal psychosocial stress on child outcomes: beyond the HPA axis. Eur Child Adolesc Psychiatry (2014) 23:943–56. doi: ten.1007/s00787-014-0566-3
PubMed Abstract | CrossRef Full Text | Google Scholar
120. Bailey MT, Lubach GR, Coe CL. Prenatal stress alters bacterial colonization of the gut in infant monkeys. J Pediatr Gastroenterol Nutr. (2004) 38:414–21. doi: x.1097/00005176-200404000-00009
PubMed Abstract | CrossRef Full Text | Google Scholar
121. Schiffrin EJ, Carter EA, Walker WA, Frieberg Due east, Benjamin J, Israel EJ. Influence of prenatal corticosteroids on bacterial colonization in the newborn rat. J Pediatr Gastroenterol Nutr. (1993) 17:271–5.
PubMed Abstract | Google Scholar
122. Wenzl HH, Schimpl K, Feierl G, Steinwender Chiliad. Event of prenatal cortisone on spontaneous bacterial translocation from alimentary canal in neonatal rat. Dig Dis Sci. (2003) 48:1171–6. doi: 10.1023/A:1023789301547
PubMed Abstruse | CrossRef Total Text | Google Scholar
125. Reid Grand, Soboh F, Bruce AW, Mittelman M. Outcome of nutrient composition on the in vitro growth of urogenital lactobacilli and uropathogens. Tin J Microbiol. (1998) 44:866–71. doi: 10.1139/w98-068
PubMed Abstruse | CrossRef Full Text | Google Scholar
126. Rousseau 5, Lepargneur JP, Roques C, Remaud-Simeon M, Paul F. Prebiotic effects of oligosaccharides on selected vaginal lactobacilli and pathogenic microorganisms. Anaerobe (2005) 11:145–53. doi: 10.1016/j.anaerobe.2004.12.002
PubMed Abstract | CrossRef Full Text | Google Scholar
127. Hütt P, Lapp Eastward, Štšepetova J, Smidt I, Taelma H, Borovkova N, et al. Characterisation of probiotic backdrop in human vaginal lactobacilli strains. Microb Ecology Wellness Dis. (2016) 27:30484. doi: 10.3402/mehd.v27.30484
PubMed Abstruse | CrossRef Full Text | Google Scholar
128. Saduakhasova S, Kushugulova A, Shakhabayeva G, Kozhakhmetov S, Khasenbekova Z, Tynybayeva I, et al. Lactobacillus for vaginal microflora correction. Cent Asian J Glob Health (2014) 3(Suppl):171. doi: 10.5195/CAJGH.2014.171
PubMed Abstract | CrossRef Full Text | Google Scholar
130. Tester R, Al-Ghazzewi FH. Intrinsic and extrinsic carbohydrates in the vagina: a short review on vaginal glycogen. Int J Biol Macromol. (2018) 112:203–6. doi: 10.1016/j.ijbiomac.2018.01.166
PubMed Abstract | CrossRef Full Text | Google Scholar
133. Otsuki K, Tokunaka K, Oba T, Nakamura One thousand, Shirato Due north, Okai T. Assistants of oral and vaginal prebiotic lactoferrin for a woman with a refractory vaginitis recurring preterm delivery: appearance of lactobacillus in vaginal flora followed past term delivery. J Obstet Gynaecol Res. (2014) forty:583–5. doi: 10.1111/jog.12171
PubMed Abstract | CrossRef Full Text | Google Scholar
134. Giunta G, Giuffrida Fifty, Mangano K, Fagone P, Cianci A. Influence of lactoferrin in preventing preterm delivery: a pilot written report. Mol Med Rep. (2012) 5:162–vi. doi: 10.3892/mmr.2011.584
PubMed Abstruse | CrossRef Full Text | Google Scholar
136. Schmitz T, Souil E, Herve R, Nicco C, Batteux F, Germain G, et al. PDE4 inhibition prevents preterm delivery induced by an intrauterine inflammation. J Immunol. (2007) 178:1115–21. doi: ten.4049/jimmunol.178.2.1115
PubMed Abstract | CrossRef Full Text | Google Scholar
hardtsplaccut1946.blogspot.com
Source: https://www.frontiersin.org/articles/10.3389/fendo.2018.00568/full
0 Response to "How Does Stress Affect Genitourinary System Peer Reviewed"
Post a Comment